Antifibrosis Two-dimensional and Three-dimensional Quantitative Structureactivity Relationship Studies on N1-substituted Phenylhydroquinolinone Derivatives
M. Abul Kashem Liton*, M. Idrish Ali
DOI:
Volume 4, Issue 4 | Pages: 374-385
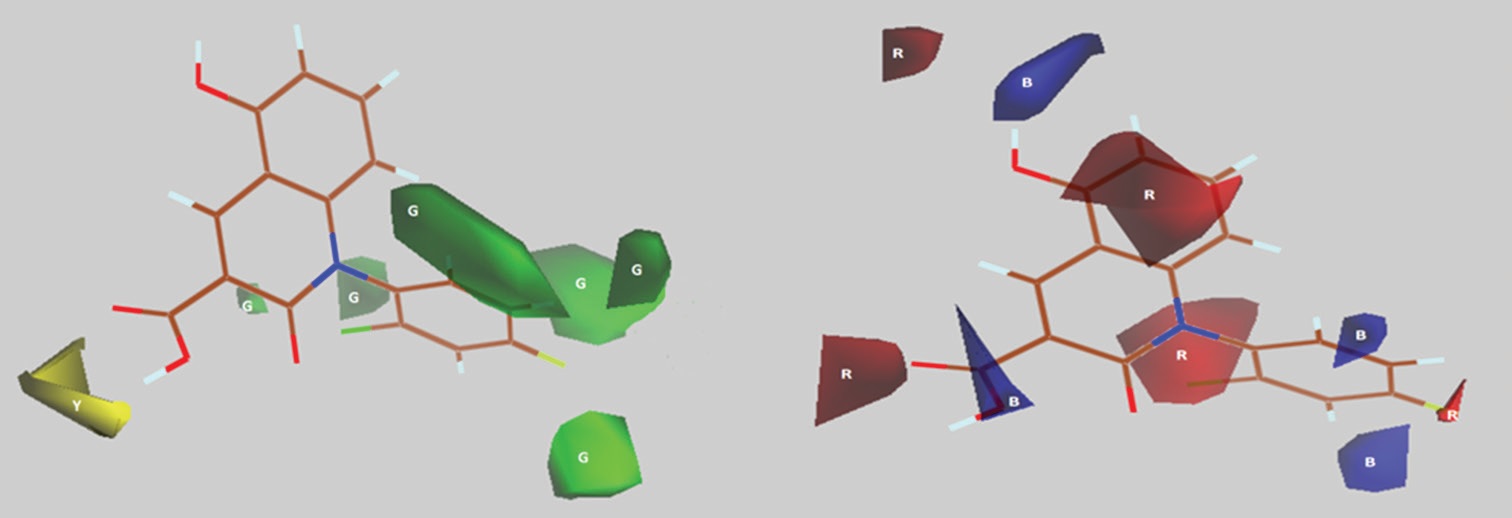
Abstract
A series of 35 N1-substituted phenylhydroquinolinone derivatives were subjected to two-dimensional (2D)- and
three-dimensional quantitative structure-activity relationship (3D-QSAR) studies applying multiple linear
regression (MLR), principal component regression (PCR), partial least square regression (PLSR), artificial neural
network (ANN), and molecular interaction energy fields (MIFs) analyses. The best predictive model obtained by
MLR, PCR, PLSR, ANN for 2D-QSAR, and MIFs at 2.0 Å grids spacing for 3D-QSAR gave the cross-validated
correlation coefficient q2 of 0.589, 0.538, 0.534, 0.815, and 0.590 and squared correlation coefficient r2 of 0.863,
0.848, 0.791, 0.954, and 0.940, respectively. The statistically significant models were established from a training
set of 28 molecules, which were validated by evaluation of test set of 7 molecules. The predicted antifibrosis
activities showed a very good agreement with experimental values in both 2D- and 3D-QSAR studies.
Keywords
Multiple linear regression Principal component regression Partial least square regression Artificial neural network Molecular interaction energy fields Leave one out.References
No references available for this article.
Citation
M. Abul Kashem Liton*, M. Idrish Ali. Antifibrosis Two-dimensional and Three-dimensional Quantitative Structureactivity Relationship Studies on N1-substituted Phenylhydroquinolinone Derivatives. Indian J. Adv. Chem. Sci. 2017; 4(4): 374-385 .