The Ground State Comparative Study of Proton Affi nities and Associated Parameters of Conjugated α,β-unsaturated Carbonyl Compounds in Gas and Aqueous Phases by Density Functional Theory Method
Biswarup Mandal, Umasankar Senapati, Bhudeb Ranjan De*
DOI:
Volume 4, Issue 4 | Pages: 401-408
Abstract
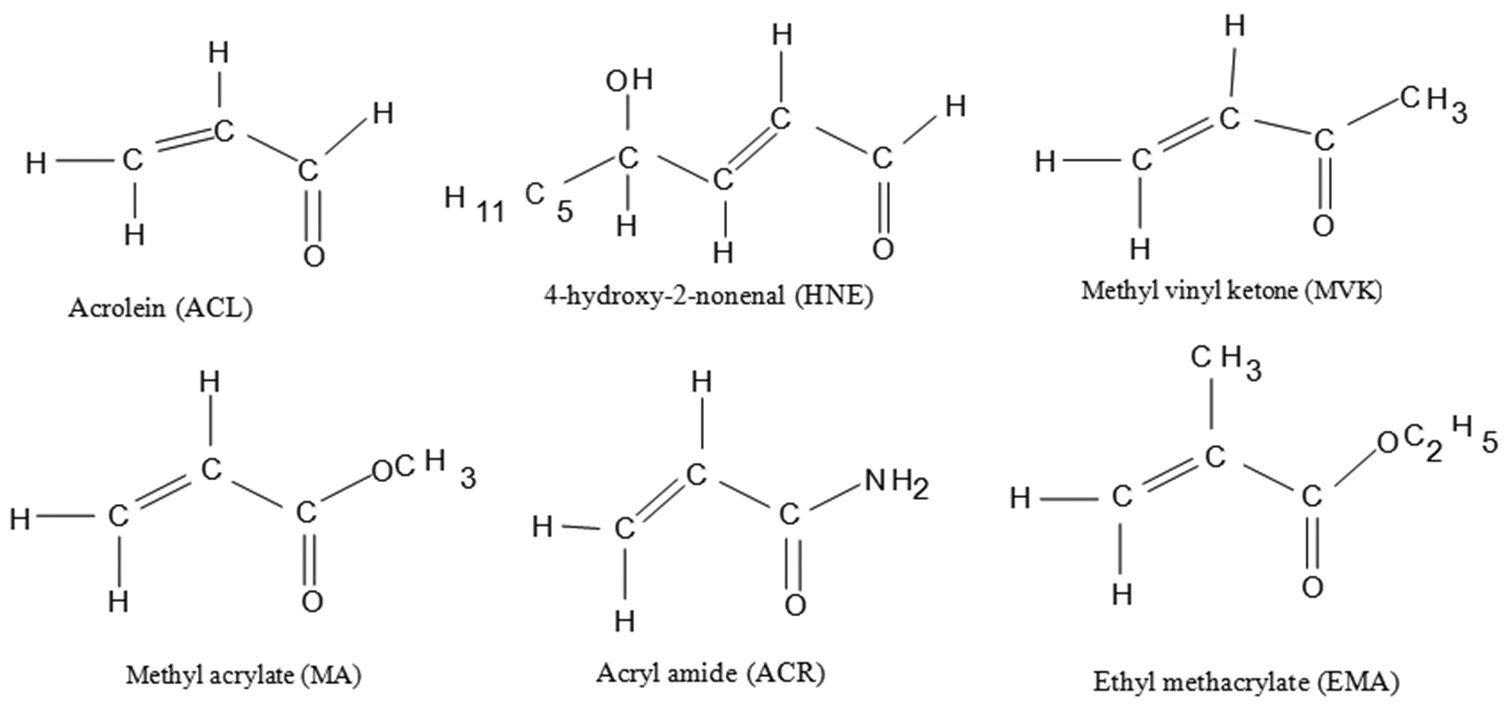
The proton affinities (PA) of a series of α,β-unsaturated carbonyl compounds (acrolein [ACL], 4-hydroxy-2-
nonenal, methyl vinyl ketone, acrylamide [ACR], methyl acrylate, and ethyl methacrylate, and their O-protonated
counterparts have been computed using density functional theory [Becke, Lee, Yang and Parr] method using
6-311G[d,p]) basis sets with complete geometry optimizations in both gaseous and aqueous phases. The
O-protonation in both phases is observed to be exothermic, and the stereochemical disposition of proton is
observed to be almost equal in each case. PA values are affected due to the presence of different length of alkyl
chain and the different substituent at carbonyl carbon. In gas phase, PA of ACR is maximum, whereas it is minimum
in ACL. In aqueous phase, the PA of the carbonyl compounds decrease in the order as –H>–NH2>–CH3>–
OC2H5>–OCH3 substituent at carbonyl carbon. Atom electron density is recorded by natural population analysis
along with Mulliken net charge. A proper correlation of PA with a number of computed system parameters like net
charge on the carbonyl oxygen of unprotonated and protonated bases, charge on proton of protonated bases, and
also the computed hardness (η) of the unprotonated bases in both phases have been explained thoroughly. The
overall basicities are explicated considering the contribution from carbonyl group and distant atom.
Keywords
Unsaturated Natural population analysis Aqueous Becke; Lee; Yang and Parr Density functional theory.References
No references available for this article.
Citation
Biswarup Mandal, Umasankar Senapati, Bhudeb Ranjan De*. The Ground State Comparative Study of Proton Affi nities and Associated Parameters of Conjugated α,β-unsaturated Carbonyl Compounds in Gas and Aqueous Phases by Density Functional Theory Method. Indian J. Adv. Chem. Sci. 2017; 4(4):401-408.