The electrochemical redox behavior of Pyro catechol violet and its quantitative measurement at graphene modified glassy carbon electrode: A voltammetric study
G. V. Raghunatha Reddy, T. Madhusudana Reddy*, P. Gopal and P. V. Narayana
DOI:
Volume 4, Issue 4 | Pages: 490-495
Abstract
The electrochemical oxidation and reduction behavior of pyrocatechol violet (PCV) at graphene modified glassy
carbon electrode were investigated by employing cyclic voltammetry (CV) and differential pulse voltammetry
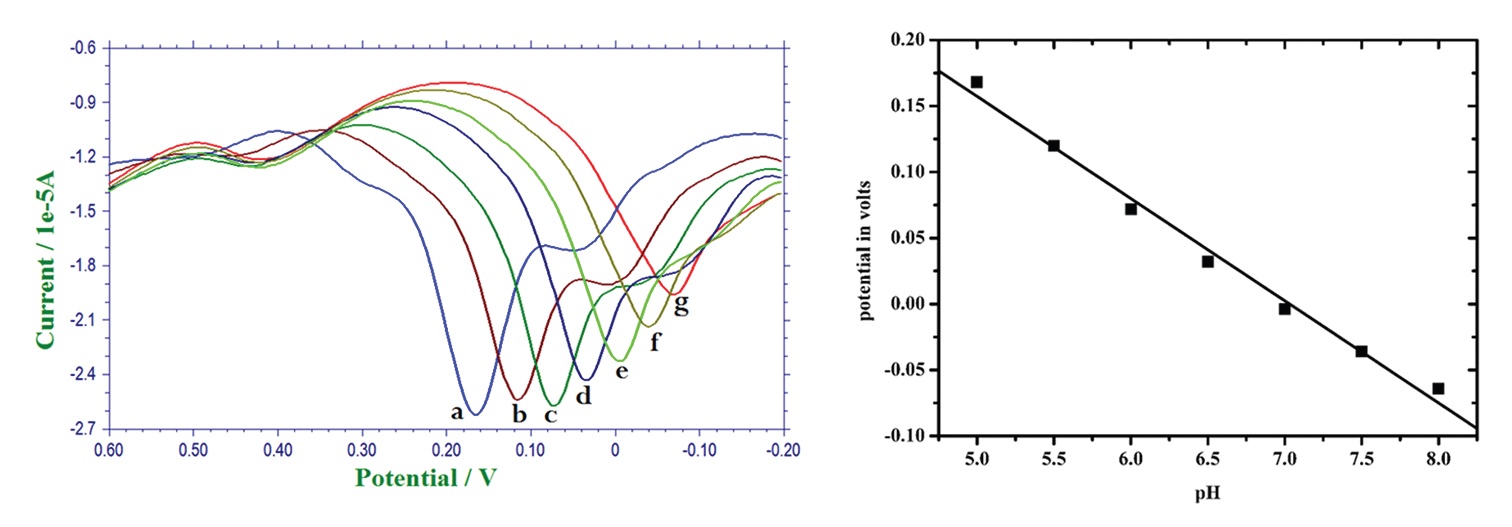
techniques. PCV exhibited an oxidation peak at ≈ 0.16 V and a reversible reduction peak at 0.14 V. The effect
of pH on the PCV was observed using the phosphate buffer solution, and pH-5.0 was chosen as optimum pH
for all the remaining experiments. The effect of scan rate on the PCV was observed and it was determined as
diffusion controlled process. We have also studied, the effect of PCV concentration on the peak current response,
from the linear plot of the calibration curve, we have evaluated the limit of detection and limit of quantification
as 3.248 × 10−8 M and 10.22 × 10−8 M. Finally, the surface nature of the electrode was characterized using the
electrochemical impedance spectroscopy.
Keywords
Pyrocatechol violet Cyclic voltammetry Differential pulse voltammetry Electrochemical impedance spectroscopy Glassy carbon electrode.References
No references available for this article.
Citation
G. V. Raghunatha Reddy, T. Madhusudana Reddy*, P. Gopal and P. V. Narayana. The electrochemical redox behavior of Pyro catechol violet and its quantitative measurement at graphene modified glassy carbon electrode: A voltammetric study. Indian J. Adv. Chem. Sci. 2017; 4(4):490-495 .