Study on Interactions of Some Metal Perchlorates Prevailing in Formamide by Physicochemical Approach
Pritam De, Mahendra Nath Roy
DOI:
Volume 3, Issue 2 | Pages: 147-154
Abstract
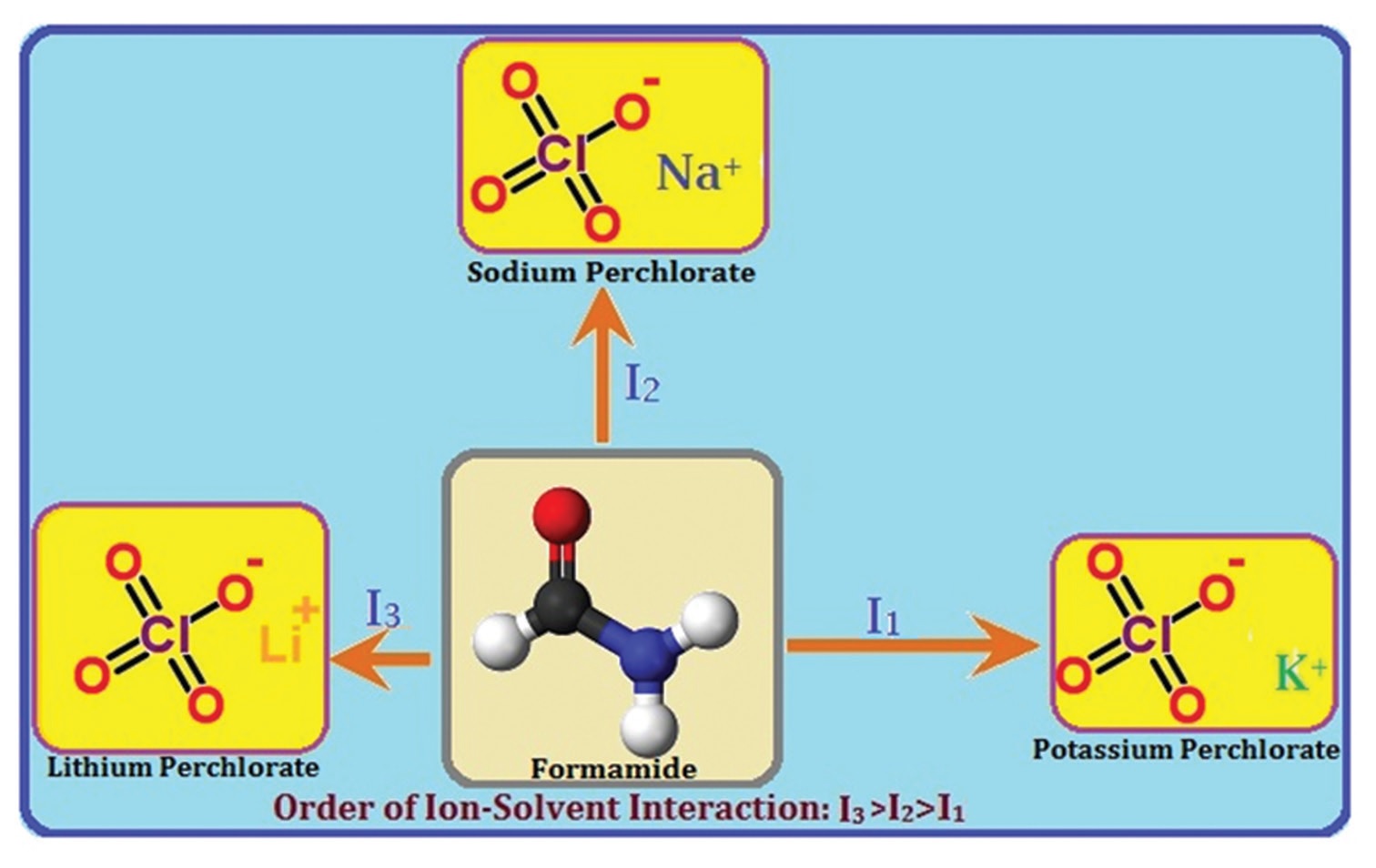
The Physicochemical properties, electrolytic conductivities (Λ), densities (r), viscosities (h) and speed of sound
(u) of lithium perchlorate, sodium perchlorate, and potassium perchlorate have been evaluated in pure formamide
at 298.15 K. Association constants (KA), limiting molar conductances (Λ0), and co-sphere diameter (R) for ion-
pair formation have been obtained from Fuoss conductance equation. Apparent molar volume (fV ), viscosity
B-coefficient and apparent molar adiabatic compressibility (fK) have been calculated from the measurement of
density (r), viscosity (h) and speed of sound (u), respectively. Masson Equation has been employed to inspect the
ion-solvent and ion-ion interactions from the values of limiting apparent molar volume (fV
0 ) and experimental
slope (SV
* ), respectively. Ion-ion and ion-solvent interactions have been interpreted with the help of the derived
parameters A and B obtained from Jones-Dole equations, respectively. The adiabatic compressibility (bS) and
limiting apparent molar adiabatic compressibility (fK
0 � ) have been assessed using the u values.
Keywords
Metal perchlorates Ion-solvent interaction Conductance Walden’s Product Adiabatic compressibility.References
No references available for this article.
Citation
Pritam De, Mahendra Nath Roy. Study on Interactions of Some Metal Perchlorates Prevailing in Formamide by Physicochemical Approach. Indian J. Adv. Chem. Sci. 2015; 3(2):147-154.