Study on Diverse Interactions of Vitamin Molecules Insight into H2o + [Epy] Bf4 Systems by Physicochemical Contrivance
Mahendra Nath Roy, Milan Chandra Roy, Siti Barman, Pran Kumar Roy, Kamalesh Roy
DOI:
Volume 3, Issue 3 | Pages: 204-218
Abstract
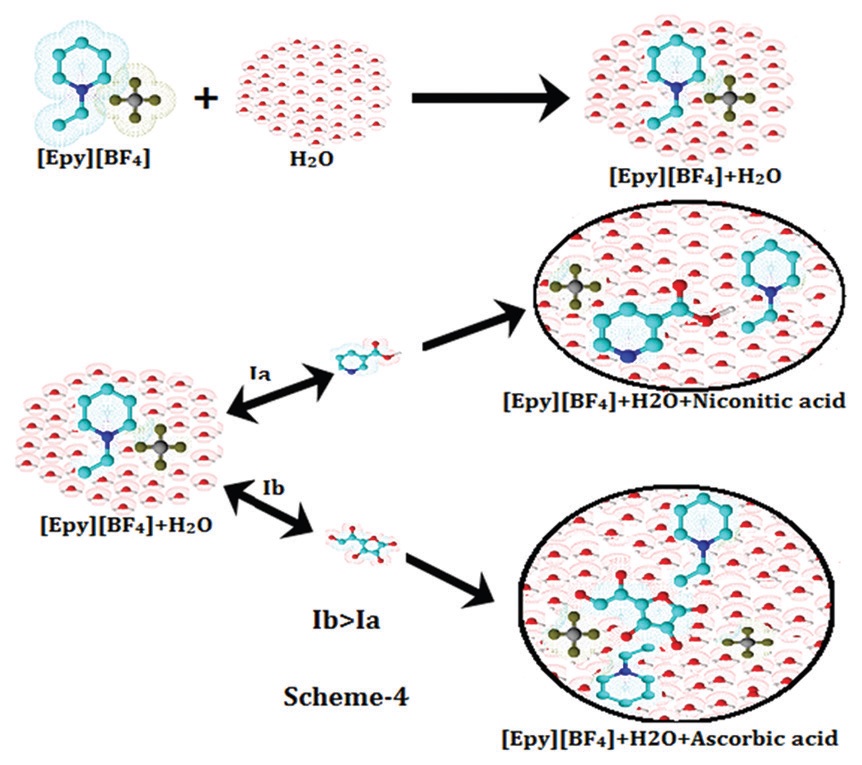
Physicochemical, such as volumetric, viscometric, refracto metric, and acoustic study of solute-solvent interactions
of nicotinic acid and ascorbic acid prevailing in aqueous solution of 1-ethyl pyridinium tetrafluoroborate
([EPy] BF4), have been reported at different temperatures. Using the density data, Masson equation has been
employed to obtain the limiting values at infinite dilution by the extrapolated to zero concentration of the
apparent molar volumes ( φV
0 ) and experimental slopes ( SV
* ), which interpreted the solute-solvent and solute-
solute interactions, respectively. Using the Jones-Dole equation, the viscosity data were analyzed to determine the
viscosity A and B- coefficient, which have also been interpreted the solute-solute and solute-solvent interaction
respectively in the solutions. The Lorentz-Lorenz equation has been employed to determine the molar refractions
(RM). The temperature dependence behavior of partial molar quantities has been determined for the vitamins. At
infinite dilution, limiting apparent molar adiabatic compressibilities ( φK
0 ) of these vitamins were evaluated and
discussed. The role of the solvent (aqueous ionic liquid mixture), and the extent of solute-solute and solute-solvent
interactions to the solution complexes have also been analyzed and discussed through the derived properties.
Keywords
Apparent molar volume Solute-solvent interaction Solute-solute interaction Vitamins Ionic Liquid.References
No references available for this article.
Citation
Mahendra Nath Roy, Milan Chandra Roy, Siti Barman, Pran Kumar Roy, Kamalesh Roy. Study on Diverse Interactions of Vitamin Molecules Insight into H2o + [Epy] Bf4 Systems by Physicochemical Contrivance. J Appl Pharm Sci. 2015; 3(3):204-218.