Volumetric and Viscometric Behavior of Ferrous Sulfate in Aqueous Lactose Solutions at Different Temperatures
Abhijit Sarkar, Bijan Kumar Pandit, Kaushik Acharjee, Biswajit Sinha
DOI:
Volume 3, Issue 3 | Pages: 219-229
Abstract
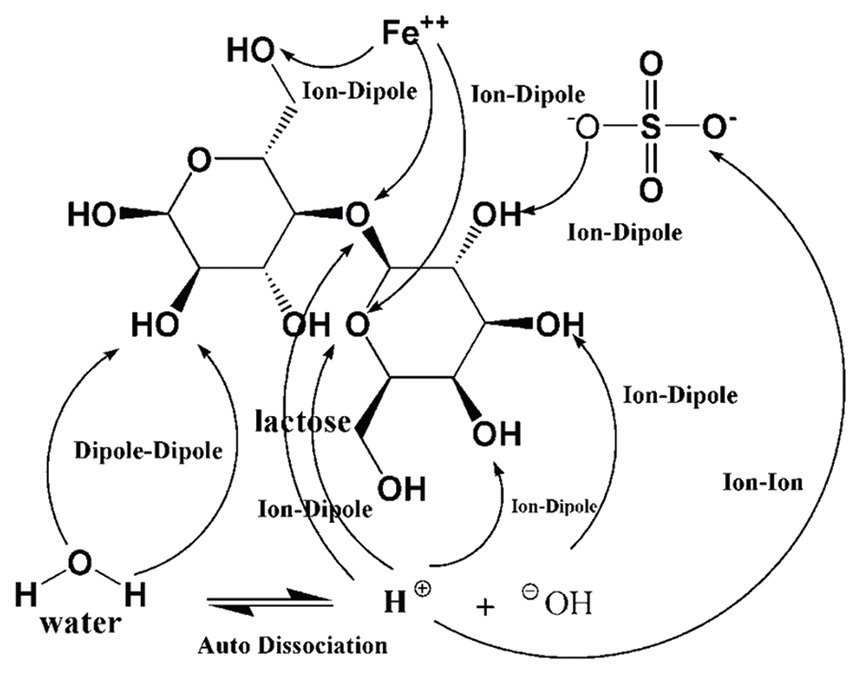
Apparent molar volumes and viscosity B-coefficients for Ferrous sulfate in aqueous lactose solutions were
determined from solution density and viscosity measurements at (298.15, 303.15, 308.15, 313.15) K as a function
of FeSO4 concentration. The standard partial molar volume and slopes obtained from the Masson equation have
been interpreted in terms of solute-solvent and solute-solute interactions, respectively. The viscosity data were
analyzed using the Jones-Dole equation and the derived parameters A and B were interpreted in terms of solute-
solute and solute-solvent interactions respectively. The standard volume of transfer and viscosity B-coefficients
of transfer of ferrous sulfate from water to aqueous lactose solutions were derived to study various interactions
in the ternary solutions. The activation parameters of viscous flow for the ternary solutions were also calculated
and explained in terms of transition state theory.
Keywords
Partial molar volumes Viscosity B-coefficients Ferrous sulfate Aqueous lactose solutions.References
No references available for this article.
Citation
Abhijit Sarkar, Bijan Kumar Pandit, Kaushik Acharjee, Biswajit Sinha. Volumetric and Viscometric Behavior of Ferrous Sulfate in Aqueous Lactose Solutions at Different Temperatures. J Appl Pharm Sci. 2015; 3(3):219-229.