Method Development and Validation for Quantitative Determination of Para Chloroaniline, an Impurity, in Efavirenz Active Pharmaceutical Ingredient by Liquid Chromatography - Electrospray Ionization - Mass Spectrometry/Mass Spectrometry
Dokku Raghava Rao, Vudagandla Sreenivasulu, B. N. Uma Maheswari, M. Veera Narayana Reddy, Nadavala Siva Kumar, Abburi Krishnaiah
DOI:
Volume 3, Issue 3 | Pages: 267-273
Abstract
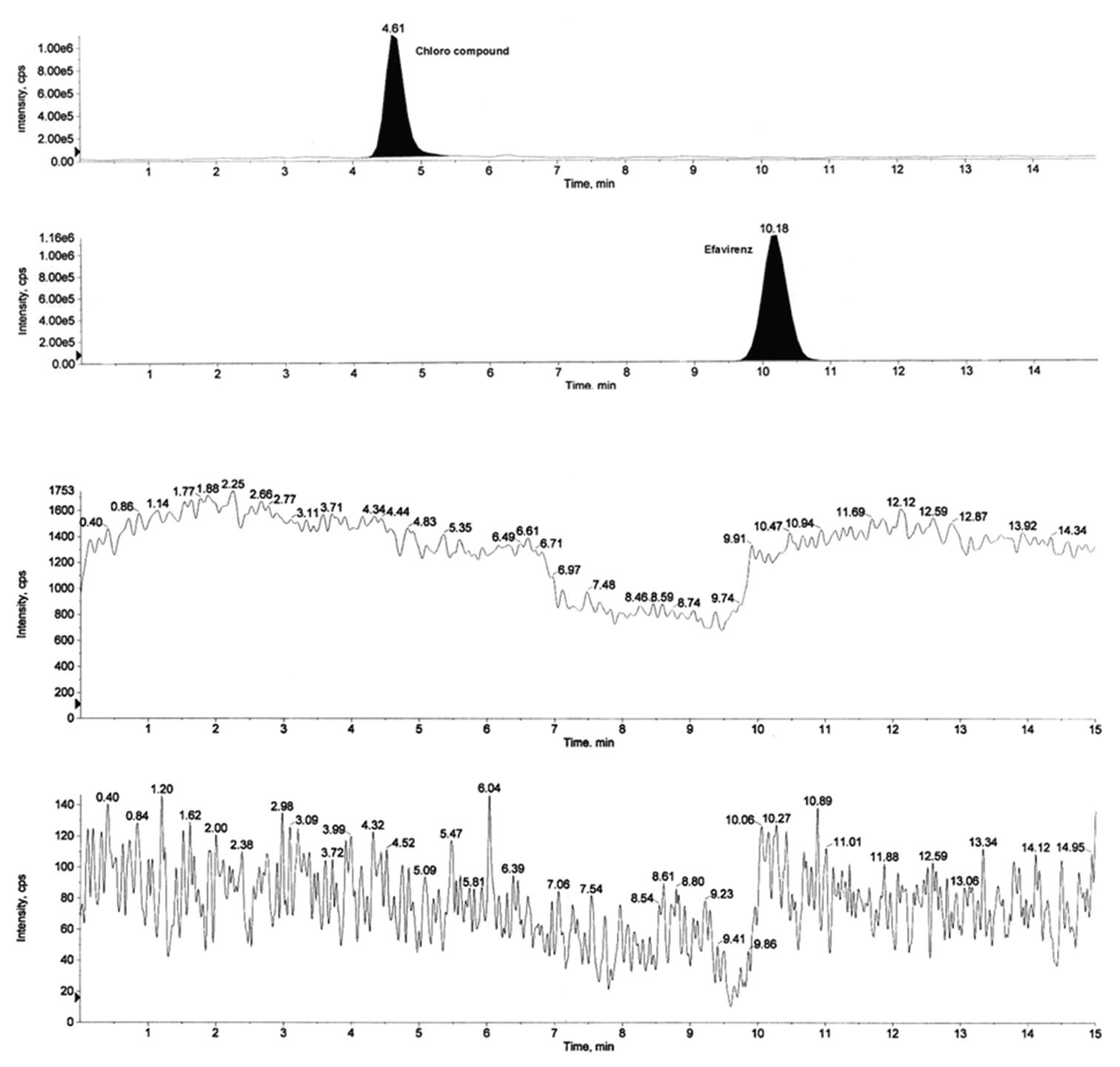
A simple, sensitive and rapid liquid chromatography (LC) - electrospray ionization (ESI) - mass spectrometry
(MS)/MS method has been developed and validated for the trace analysis (>1 ppm level) of para chloroaniline,
a genotoxic impurity, in Efavirenz drug. The chromatographic separation was achieved on a hypersil
BDS (150 × 4.6 mm, 5 μm) column using a mobile phase consisting of 5 mM ammonium acetate buffer (pH 4) and
acetonitrile (60:40, v/v) at fl ow rate of 0.7 mL/min and elution was monitored at 305 nm. The active pharmaceutical
ingredient-4000 LC-MS/MS was operated on an ESI equipped with an ESI interface operated in positive ionization
(single reaction monitoring) mode and it is able to quantitate up to 0.3 ppm of para chloroaniline. The newly
developed method was validated as per ICH guidelines.
Keywords
Efavirenz Para chloroaniline method validation Liquid chromatography-electrospray ionization-Mass Spectrometry/Mass Spectrometry Trace analysis.References
No references available for this article.
Citation
Dokku Raghava Rao, Vudagandla Sreenivasulu, B. N. Uma Maheswari, M. Veera Narayana Reddy, Nadavala Siva Kumar, Abburi Krishnaiah . Method Development and Validation for Quantitative Determination of Para Chloroaniline, an Impurity, in Efavirenz Active Pharmaceutical Ingredient by Liquid Chromatography - Electrospray Ionization - Mass Spectrometry/Mass Spectrometry. J Appl Pharm Sci. 2015; 3(3):267-273.