Study of Solvation Consequences of 1-Butyl-1-Methylpyrrolidinium Bromide and Chloride Prevailing in Protic Solvent System
Mahendra Nath Roy*, Kanak Roy, Mitali Kundu, Biraj Kumar Barman
DOI:
Volume 3, Issue 4 | Pages: 304-314
Abstract
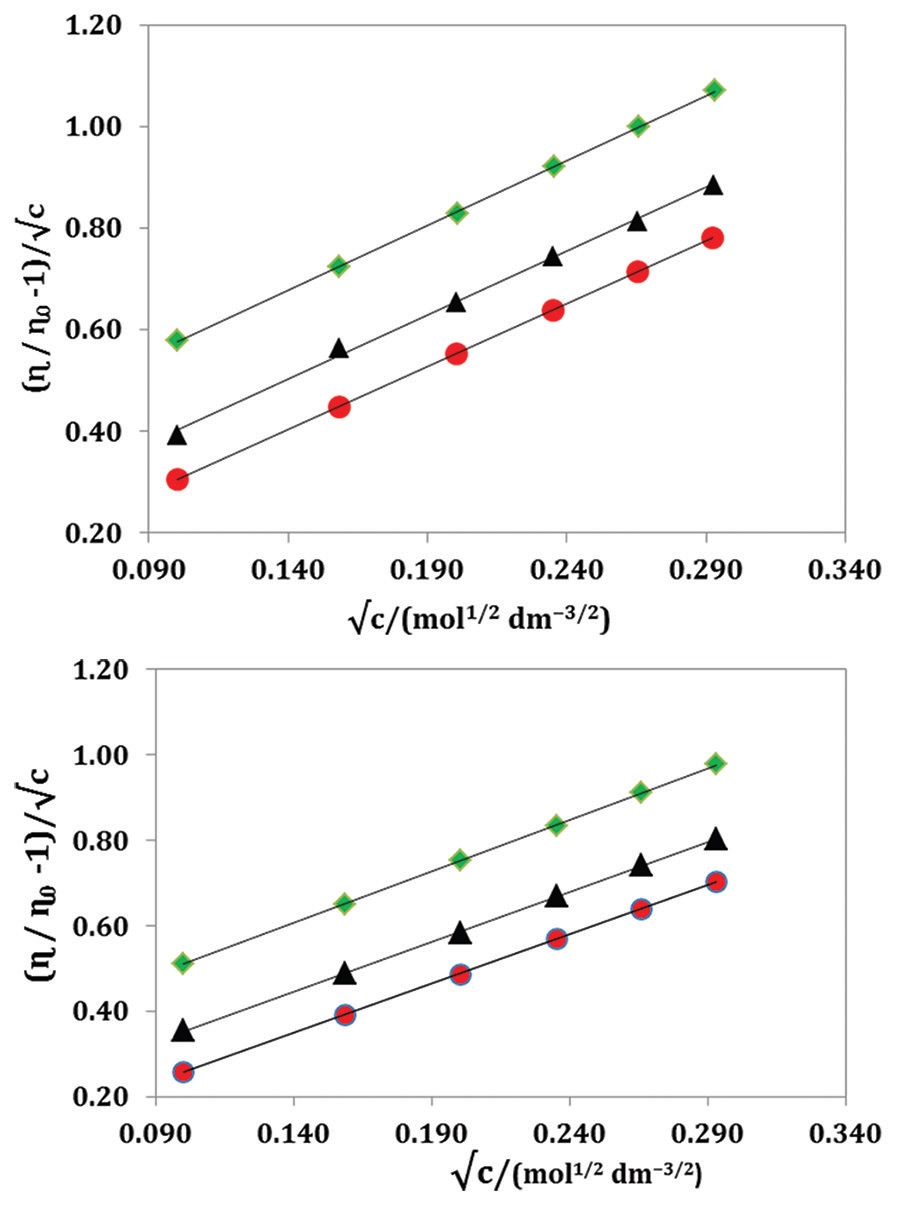
Electrolytic conductivities, densities, viscosities, and Fourier transform infrared (FTIR) studies of 1-buty-1-methyl
pyrrolidiniumbromide ([BMPyrr][Br]) and 1-butyl-1-methylpyrrolidiniumchloride ([BMPyrr][Cl]) have been
studied in methanol at different temperatures. The limiting molar conductivities, association constants, and the
distance of closest approach of the ions have been analyzed using the Fuoss conductance equation (1978) for ion-
pair formation. Ion-solvent interactions have been interpreted in terms of apparent molar volumes and viscosity
B-coefficients which are obtained from the results of density and viscosity measurements. The limiting apparent
molar volumes, experimental slopes derived from the Masson equation and viscosity A and B coefficients using
the Jones–Dole equation have been interpreted in terms of ion-ion and ion-solvent interactions respectively. The
FTIR spectra for the methanol, as well as the ionic liquids in methanol have also been recorded. The experimental
results have been discussed in terms of ion-dipole interactions, hydrogen bonds formation, structural aspect, and
configurational theory.
Keywords
Solvation consequence Ion-pair formation Viscosity Density Conductance Fourier transform infrared spectra.References
No references available for this article.
Citation
Mahendra Nath Roy*, Kanak Roy, Mitali Kundu, Biraj Kumar Barman. Study of Solvation Consequences of 1-Butyl-1-Methylpyrrolidinium Bromide and Chloride Prevailing in Protic Solvent System. Indian J. Adv. Chem. Sci. 2016; 3(4):304-314.