Development and Validation of Ultraviolet-Spectrophotometric Method for the Determination of Tamsulosin
G. Dilli Rani, C. Narasimha Rao, C. Narasimha Rao, A. Narayana, P. Venkateswarlu*
DOI:
Volume 3, Issue 4 | Pages: 323-327
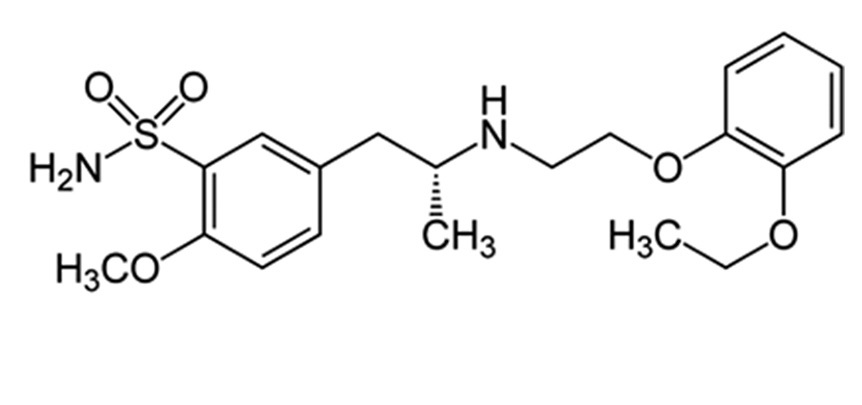
Abstract
A simple, specific, and economical ultraviolet-spectrophotometric method has been developed for the
determination of tamsulosin in pharmaceutical and biological fluid samples. Tamsulosin undergoes diazotization
when treated with the sodium nitrite and hydrochloric acid. The excess of nitrous acid during the diazotization is
removed by the addition of urea solution. The diazonium cation reacts with the coupling reagent, β-naphthol by
electrophilic substitution at the o-position of the coupling agent to produce an orange azo product. This orange
product shows maximum absorbance at 475 nm. The calibration curve is linear over the concentration range
of 25-150 mg/ml of tamsulosin. The optical characteristics of the proposed method such as molar absorptivity,
Sandell’s sensitivity, slope, and intercept were 1.5574 L.mole−1cm−1, 0.0025 μg.cm−2, 0.00394, and 0.011905 for
tamsulosin, respectively. The developed method was found to be simple, specific, robust, accurate, and precise for
the determination of tamsulosin.
Keywords
Tamsulosin Sodium nitrite Hydrochloric acid β-naphthol and ultraviolet-spectrophotometric method.References
No references available for this article.
Citation
G. Dilli Rani, C. Narasimha Rao, C. Narasimha Rao, A. Narayana, P. Venkateswarlu*. Development and Validation of Ultraviolet-Spectrophotometric Method for the Determination of Tamsulosin. Indian J. Adv. Chem. Sci. 2016; 3(4):323-327.