Development and Validation of HPLC Assay Method for the Acamprosate Ca in Commercial Tablets
C. Babu, B. Sreenivasa Rao, K.V.N. Suresh Reddy, B. Naganjaneyulu
DOI:
Volume 2, Issue 1 | Pages: 46-49
Abstract
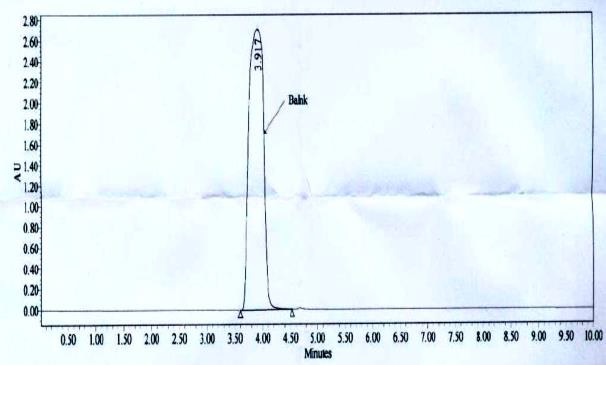
A simple, selective and accurate high performance liquid chromatography method was developed and validated
for the analysis of Acamprosate Ca in its Tablet dosage forms. Chromatographic separation was achieved
isocratically on ACE phenyl column utilizing a mobile phase of methanol and phosphate buffer (pH adjusted to
7.0 ± 0.05) with 10:90 (v/v). The flow rate is 0.8 mL min-1 and analytes are monitered with PDA detector at 210
nm. The detector response for acamprosate was linear over the concentration range from 33μg mL-1 to 528 μg
mL-1 (r2 = 0.995). The RSD of intra-day and inter-day precision were 0.42% and 0.59% respectively. The
accuracy was between 98.17% and 101.77%. There was no interference of the blank on determination of active
pharmaceutical ingredients. The LOD and LOQ values for Acamprosate were 0.33 μg mL-1 and 1.0 μg mL-1
respectively. Acamprosate was subjected to acid, alkali, oxidation and thermal degradations. The proposed
method was precise, accurate, specific and sensitive, it can be applied to the quantitative determination of drug
in tablets.
Keywords
Liquid chromatography Tablet assay Validation Acamprosate CaReferences
No references available for this article.
Citation
C. Babu, B. Sreenivasa Rao, K.V.N. Suresh Reddy, B. Naganjaneyulu. Development and Validation of HPLC Assay Method for the Acamprosate Ca in Commercial Tablets. Indian J. Adv. Chem. Sci. 2014; 2(1):46-49.