Equilibrium and Kinetic Studies for The Biosorption System of Cu(Ii) Ions From Aqueous Solution Using Eucalyptus Bark Powder
S.K. Akmal, J. Malathi, Y. Vijaya, M. Venkata Subbaia
DOI:
Volume 2, Issue 2 | Pages: 151-157
Abstract
Eucalyptus bark, an agricultural solid waste by-product has been developed into an effective and efficient
biosorbent the removal of Cu(II) from aqueous solutions. Batch biosorption experiments were carried out as a
function of pH, biosorbent dosage, contact time and initial metal ion concentration. The biosorbent Eucalyptus
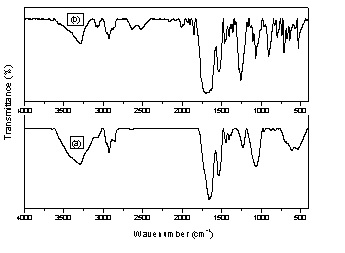
bark powder was characterized by FTIR analysis. The experimental isotherm data were analyzed using
Langmuir and Freundlich equations. The equilibrium data fit well the Langmuir isotherm. The maximum
biosorption capacity was 47.15 mg/g of Cu(II) ions onto Eucalyptus bark powder. Kinetic studies showed the
pseudo-second-order described the biosorption experimental data better than the pseudo-first-order kinetics.
The Desorption studies were carried out using dilute HCl, and the effect of HCl concentration on desorption
was studied. The finding of the present study indicates that Eucalyptus bark can be successfully used for
separation of Cu(II) from aqueous solutions.
Keywords
Biosorption; Eucalyptus bark; Cu(II); Kinetics; IsothermsReferences
No references available for this article.
Citation
S.K. Akmal, J. Malathi, Y. Vijaya, M. Venkata Subbaia. Equilibrium and Kinetic Studies for The Biosorption System of Cu(Ii) Ions From Aqueous Solution Using Eucalyptus Bark Powder. Indian J. Adv. Chem. Sci. 2014; 2(2):151-157.