Exploration of Miscellaneous Interfaces of a Green Liquid in Diverse Solvent Systems by the Process of Physicochemical Contrivances
Mahendra Nath Roy*, Biswajit Datta, Saptarshi Basak
DOI:
Volume 2, Issue 4 | Pages: 253-263
Abstract
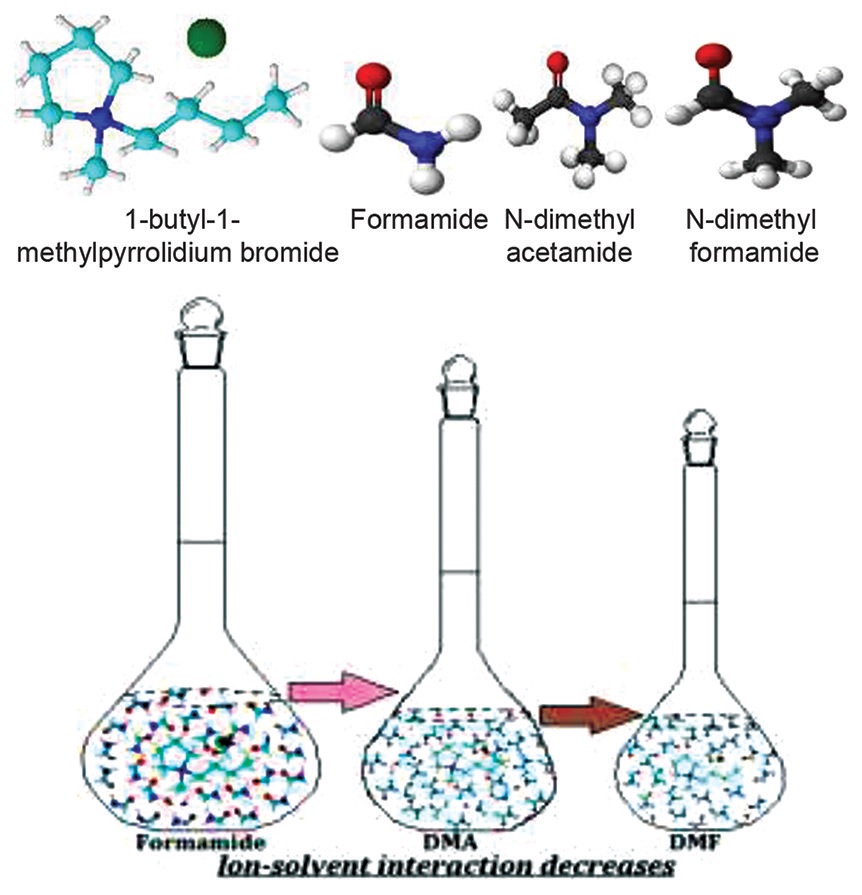
Precise measurements on electrical conductance of solutions of 1-butyl-1-methylpyrrolidium bromide in formamide,
N, N-dimethyl formamide and N, N-dimethyl acetamide have been studied at 298.15 K. The conductance data have
been analyzed by the Fuoss conductance equation (1978) in terms of limiting molar conductance, association
constant, and association diameter for ion-pair formation. The apparent molar volume, viscosity B-coefficient,
molar refraction and adiabatic compressibility have also been studied, supplemented from the values of density,
viscosity, refractive index and ultrasonic speed respectively, to interpret the ion-ion and ion-solvent interactions.
Limiting apparent molar volumes, experimental slopes have been obtained from the Masson equation. The viscosity
data have been analyzed using Jones–Dole equation to derived A and B co-efficient. Molar refractions have been
determined with the help of Lorentz–Lorenz equation and limiting apparent molar adiabatic compressibilities in
all three solvents at infinite dilution have been evaluated and discussed. Infrared study of the functional group of
solvents in presence and absences of interleukin has also been taking into account for interpreted the same.
Keywords
Ionic liquid Limiting molar conductance Ion-solvent and ion-ion interaction Viscosity B-coefficient.References
No references available for this article.
Citation
Mahendra Nath Roy*, Biswajit Datta, Saptarshi Basak. Exploration of Miscellaneous Interfaces of a Green Liquid in Diverse Solvent Systems by the Process of Physicochemical Contrivances. Indian J. Adv. Chem. Sci. 2015; 2(4):253-263.