The Weak Intermolecular Interactions in 2,6-Bis(4-Isopropylbenzylidene) Cyclohexanone: Insights from Crystallographic Studies and Semi-Empirical Quantum Chemical Calculations
S. Shalini, C. R. Girija, T.V. Venkatesha, M.M. Jotani, M. Srinivasa Murthy and K.N. Venugopala
DOI:
Volume 1, Issue 2 | Pages: 88-93
Abstract
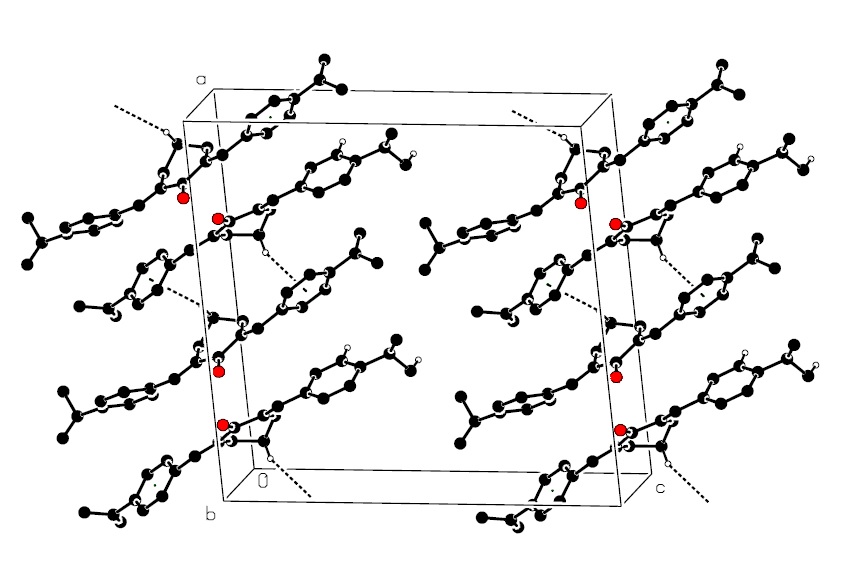
In the molecular structure of 2,6-bis(4-isopropylbenzylidene) cyclohexanone, (I), the central cyclohexane ring
adopts half-chair conformation quantified by ring puckering analysis and asymmetry parameters. The olefinic
double bonds in the title compound have E-conformation. The conformational discrepancy between the
experimental structure and theoretical one computed by AM1 semi-empirical method describes the role of short
intramolecular C-H...O contacts and a weak C-H ......interaction in the solid state of a molecule. The
parametric molecular electrostatic potential (PMEP) calculated by semi-empirical method also shows the
involvement of oxygen atom in the short intramolecular C-H... O contacts in crystal packing. The
intramolecular charge transfer resulting from HOMO-LUMO transition reflects the chemical activity of a
molecule.
Keywords
Cyclohexanone semi-empirical method parametric molecular electrostatic potential Charge transfer interactionReferences
No references available for this article.
Citation
S. Shalini, C. R. Girija, T.V. Venkatesha, M.M. Jotani, M. Srinivasa Murthy and K.N. Venugopala. The Weak Intermolecular Interactions in 2,6-Bis(4-Isopropylbenzylidene) Cyclohexanone: Insights from Crystallographic Studies and Semi-Empirical Quantum Chemical Calculations. Indian J. Adv. Chem. Sci. 2013; 1(2):88-93.