Removal of Pb(II) From Aqueous Solutions by Caesalpinia bonducella Leaf Powder(CBLP)
G. Yuvaraja, K. Pattabhi Ramaiah, M. Veera Narayana Reddy and A. Krishnaiah
DOI:
Volume 1, Issue 3 | Pages: 152-156
Abstract
Biosorption of Pb(II) ions from aqueous solutions by Caesalpinia bonducella leaf powder (CBLP) was
investigated as a function of initial pH, biosorbent dose, initial metal ion concentration and temperature. The
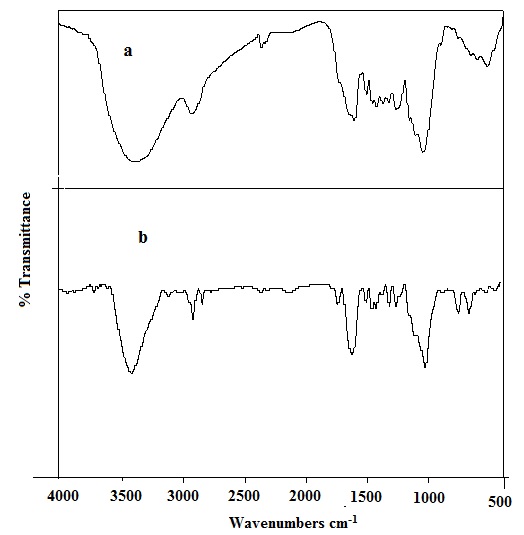
FTIR spectrum shows the involvement of different functional groups (hydroxyl, amine, carboxyl and carbonyl
groups) in Pb(II) biosorption. Kinetic data were best fitted with the pseudo-second-order kinetic model. The
batch equilibrium data were fitted well by Langmuir isotherm. The monolayer maximum biosorption capacity,
calculated from Langmuir isotherm model, was found 116.27 mg/g at 323 K.
Keywords
Biosorption; Lead; Caesalpinia bonducella leaf powder; Kinetics; IsothermsReferences
No references available for this article.
Citation
G. Yuvaraja, K. Pattabhi Ramaiah, M. Veera Narayana Reddy and A. Krishnaiah. Removal of Pb(II) From Aqueous Solutions by Caesalpinia bonducella Leaf Powder(CBLP). Indian J. Adv. Chem. Sci. 2013; 1(3):152-156.