Synthesis and Characterization of Iron(II) Coordination Complexes with PPh2Py and DPEphos Ligands: A Combined Experimental and Theoretical Study
Malabika Borah, Nabanita Saikia* , Pankaj Das
DOI: 10.22607/IJACS.2024.1203006
Volume 12, Issue 3 | Pages: 175-182
Abstract
The synthesis and characterization of Iron(II) complexes [FeCl2(η2-P,N-PPh2Py)2] (1) and [FeCl2(η2-P,P-DPEphos)2] (2), with
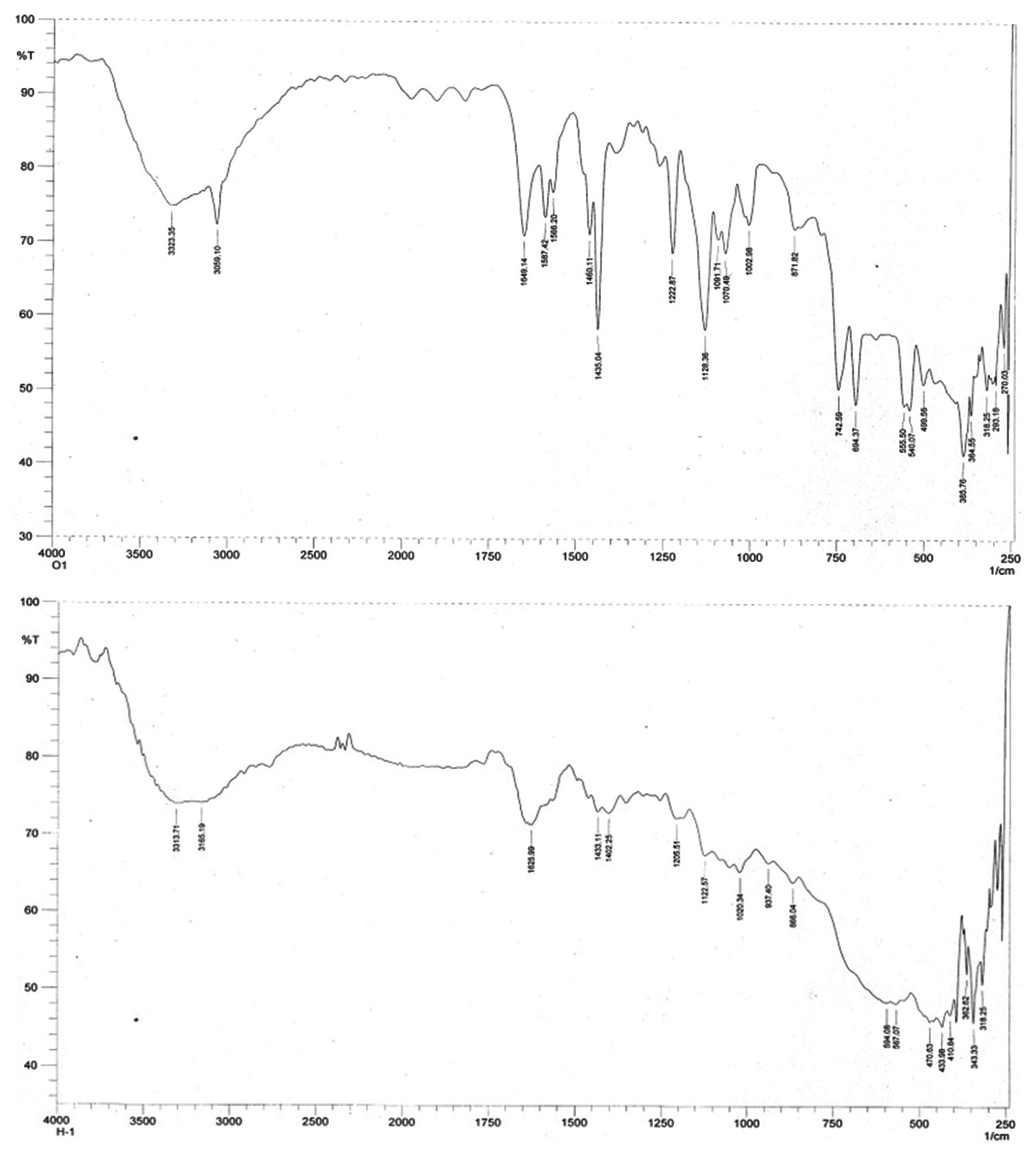
PPh2Py and DPEphos ligands, were performed using elemental analysis, ESI-mass, Fourier transform infrared spectra (FTIR),
ultraviolet-visible, 1H and 31P{1H} nuclear magnetic resonance spectroscopy. FTIR measurements predicted cis-isomer to be
the most stable form of complex 1 and trans-isomer to be the most stable form of complex 2. Quantum chemical calculations
using first-principles density functional theory were performed on the two complexes at the B3LYP/LANL2DZ/6-31+G(d,p)
level of theory. Theoretical calculations predicted that the ground state of the complexes would be a quintet spin state. However,
in complex 1, the quintet spin state led to a significant elongation in Fe–P bond length to ~3.55 Å. Thus, a singlet (S = 1/2)
spin state was considered for complex 1 which showed reasonable agreement with calculated geometric parameters. Trans-
configuration of complex 2 was shown to have a higher highest occupied molecular orbital-lowest unoccupied molecular orbital
energy gap (higher stability) than complex 1 which was attributed to the nature of the ligand coordinated to Fe(II) ion.
Keywords
Fe(II) complexes PPh2Py and DPEphos ligands elemental analysis Fourier Transform Infrared spectra NMR spectroscopy Density Functional Theory.References
No references available for this article.
Citation
Malabika Borah, Nabanita Saikia* , Pankaj Das. Synthesis and Characterization of Iron(II) Coordination Complexes with PPh2Py and DPEphos Ligands: A Combined Experimental and Theoretical Study. Indian J. Adv. Chem. Sci. 2024; 12(3):175-182 .