Nickel Borate Mediated Synthesis of Imidazole-Based Heterocycles
Vishwa Deepak Tripathi
DOI: DOI: 10.22607/IJACS.2025.1304001
Volume 13, Issue 4 | Pages: 184-189
Abstract
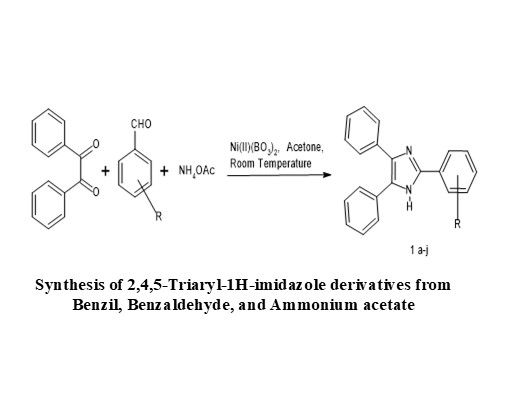
The synthesis of 2,4,5-triaryl-1H-imidazole derivatives has been achieved efficiently through a straightforward and versatile multicomponent reaction strategy. In this method, Nickel (II) borate acts as a highly effective catalyst in acetone medium, promoting the one-pot condensation of a 1,2-dicarbonyl compound, an aromatic aldehyde, and ammonium acetate, which serves as the in-situ ammonia donor. This catalytic system provides a convenient and environmentally benign alternative to conventional protocols, minimizing the need for harsh reaction conditions or complex purification steps. The reaction proceeds smoothly with good compatibility toward a variety of aromatic aldehydes, thereby furnishing the desired triaryl-imidazole scaffolds in moderate to excellent yields within a relatively short reaction time. The advantages of this methodology include operational simplicity, mild conditions, low cost of catalyst, and reproducibility, making it a practical approach for the synthesis of imidazole-based heterocycles. Such compounds are of significant interest due to their broad applications in medicinal chemistry and materials science.
Keywords
2 4 5-Triaryl-1H-imidazole multicomponent reaction Nickel (II) borate simple workup.References
No references available for this article.
Citation
Vishwa Deepak Tripathi. Nickel Borate Mediated Synthesis of Imidazole-Based Heterocycles. Indian J. Adv. Chem. Sci. 2026; 13(4): 184-189.