Rapid Bromination of some Regioisomers of Dimethylphenol by Molecular Bromine in Aqueous Solution: Kinetic Verification of Reactivities
Rashmi D. Katkar, Sushil B. Kapoor
DOI: DOI: 10.22607/IJACS.2025.1401004
Volume 14, Issue 1 | Pages: 31-35
Abstract
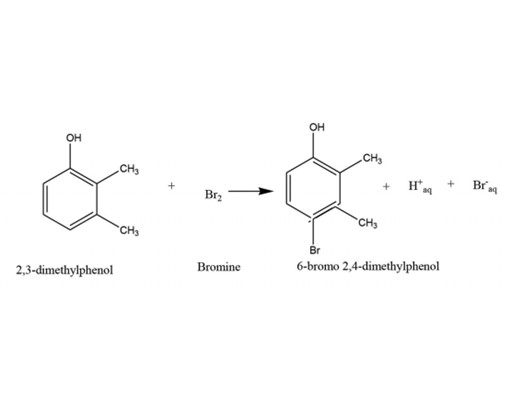
Kinetics of bromination of 2,3-dimethylphenol, 2,4-dimethylphenol, and 2,5-dimethylphenol by molecular bromine in aqueous
solution at pH 7.0 have been studied. The Kinetic data obtained conclusively enlightened the relative reactivities of these isomers
quantitatively. All three reaction studies were found to be rapid and hence needed a special technique to follow the kinetics.
In the present study, a rotating platinum electrode is used and yielded specific reaction rates ranging from 1.32 × 103
M−1S−1 to 2.34 × 103 M−1S−1, activation energies from 30.8 KJ mole−1 to 49.1 KJ mole−1 and frequency factor from 5.85 × 108
M−1S−1 to 5.32 × 1011 M−1S−1 at 25°C and 7.0 pH. Stereochemical principles justifies this observed reactivities of three isomers under
study. Thus, the speculated stereochemistry of these isomers was quantitatively verified using Kinetic as a tool.
Keywords
Bromination Dimethylphenols Kinetics Molecular bromine Rotating platinum electrode StereochemistryReferences
No references available for this article.
Citation
Rashmi D. Katkar, Sushil B. Kapoor. Rapid Bromination of some Regioisomers of Dimethylphenol by Molecular Bromine in Aqueous Solution: Kinetic Verification of Reactivities. Indian J. Adv. Chem. Sci. 2026; 14(1):31-35.