Synthesis, Characterization, Density Functional Theory, Molecular Docking Studies, and Biological Activities of 6,8-dichloro-3-phenyl[1,2,4]triazolo[3,4-b] [1,3]benzoxazole Derivatives
N. D. Jayanna*, T. Manjuraj, Mohammed Imadadulla
DOI: 10.22607/IJACS.2024.1203010
Volume 12, Issue 3 | Pages: 204-211
Abstract
Triazole and benzoxazole nucleus are the most adorable moieties in the field of medicinal chemistry. In our prior research, we
employed synthesis of target molecules bearing both active nucleuses of triazole and benzoxazole derivatives. The compounds
6,8-dichloro-3-phenyl[1,2,4]triazolo[3,4-b][1,3]benzoxazole derivatives (4-11) were synthesized by reacting 5,7-Dichloro-2-
hydrazino-1,3-benzoxazole (3) with various compounds containing carboxylic groups through conventional method. The target
molecules have been characterized by infrared, 1H NMR, 13C NMR, and mass spectral studies. The synthesized molecules
subjected for antibacterial, insecticidal, and antioxidant activities to know the efficacy of molecules. In addition to this
computational density functional theory using B3LYP/LANL2DZ method in the ground state was performed. The calculations
were done to confirm the geometry of the molecules and also the highest occupied molecular orbital-lowest unoccupied
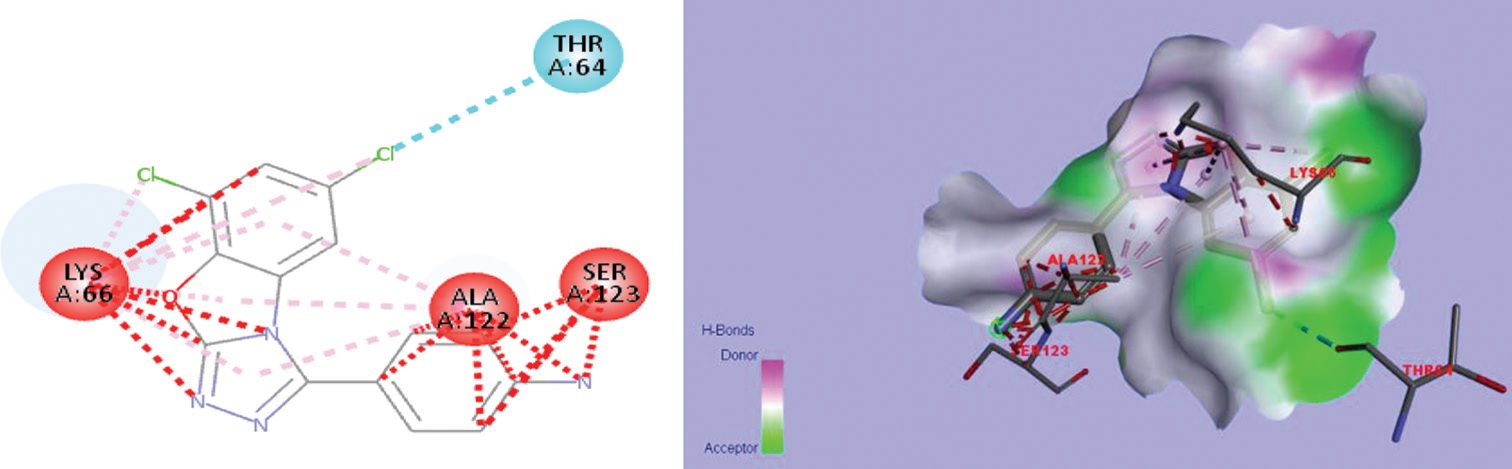
molecular orbital excitation energy levels also calculated. Further, in addition to this, molecular docking studies were performed
with protein receptor PDB: 3MNG exhibits the highest binding interactions with all the compounds.
Keywords
Benzoxazole Density functional theory Highest occupied molecular orbital Insecticidal activity Lowest unoccupied molecular orbital Molecular docking and Triazole.References
No references available for this article.
Citation
N. D. Jayanna*, T. Manjuraj, Mohammed Imadadulla. Synthesis, Characterization, Density Functional Theory, Molecular Docking Studies, and Biological Activities of 6,8-dichloro-3-phenyl[1,2,4]triazolo[3,4-b] [1,3]benzoxazole Derivatives. Indian J. Adv. Chem. Sci. 2024; 12(3): 204-211 .