Organocatalyst-Promoted [3+2] Cycloaddition Reaction of Azomethine Ylides and Olefinic Dipolarophiles
Siyaram Prasad*
DOI: 0.22607/IJACS.2023.1102008
Volume 11, Issue 2 | Pages: 115-117
Abstract
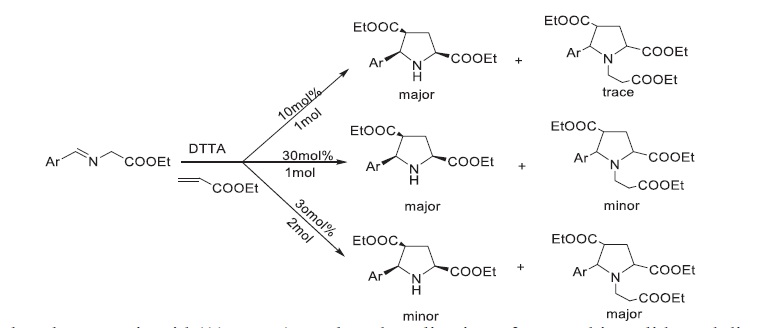
An efficient enantio- and diastereoselective synthesis of substituted pyrrolidines have been reported using tartaric acid as an
organocatalyst through [3+2] cycloaddition of azomethine ylides (N-alkylidene glycine esters) and diploarophiles in good-
to-excellent yields. We have also used di-p-toluoyl-L-tartaric acid (DTTA) (30% mmol) as an organocatalyst which gives
[3+2]-dipolar cycloaddition product along with aza-Michael product by the reaction of dipolarophiles on cycloaddition product.
However, DTTA (5% mmol) afforded cycloaddition product as a major product.
Keywords
Organocatalysis Cycloaddition Dipolarophiles Micheal reaction Ylides.References
No references available for this article.
Citation
Siyaram Prasad*. Organocatalyst-Promoted [3+2] Cycloaddition Reaction of Azomethine Ylides and Olefinic Dipolarophiles. Indian J. Adv. Chem. Sci. 2023; 11(2):115-117.