Structure and Electronic Features of Radicals: Detection and Characterization
Sayantan Mondal*
DOI: 10.22607/IJACS.2023.1103001
Volume 11, Issue 3 | Pages: 158-161
Abstract
Radical reactions occur through the intermediacy of odd electron species and are among the key fundamental classes of organic
transformations. Such processes play important roles in mechanistic and synthetic organic chemistry and are essential for many
biological and materials applications. In this review, structure and electronic features of radicals of organic molecules have been
discussed in details detection and characterization procedure.
Keywords
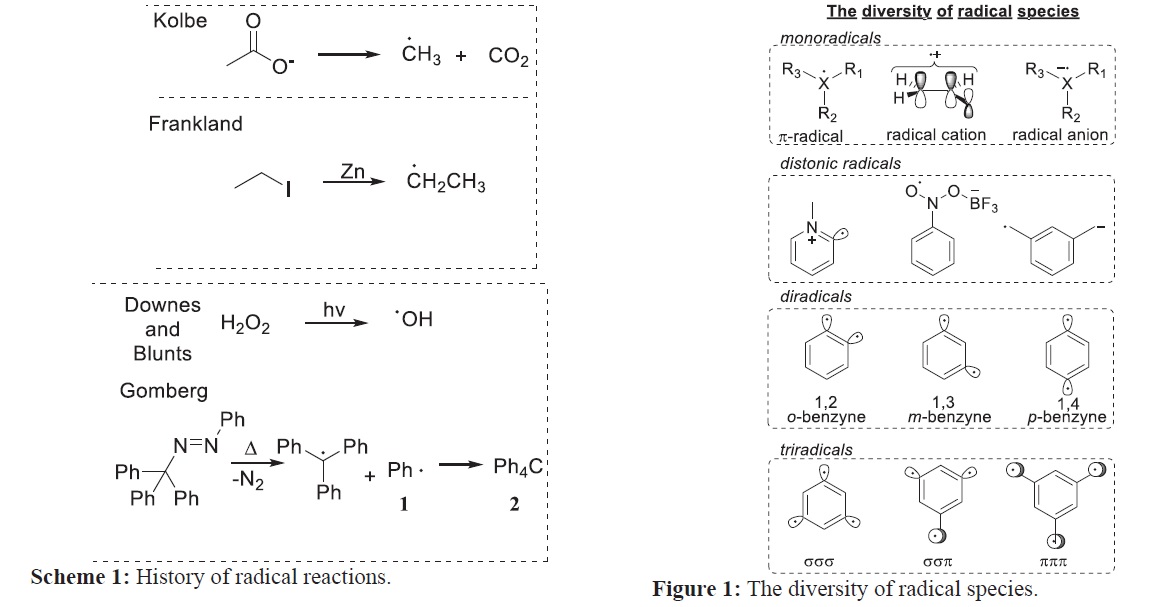
Radical Intermediate Molecular orbital Spectroscopy. Indian Journal of Advances in Chemical Science Article *Corresponding author: Sayantan Mondal E-mail: sayantaniitkgp2006@gmail.com ISSN NO: 2320-0898 (p); 2320-0928 (e) DOI: 10.22607/IJACS.2023.1103001 Received: 14th April 2023; Revised: 10th May 2023; Accepted: 21st June 2023 KROS Publications 159 www.ijacskros.comIndian Journal of Advances in Chemical Science 2023; 11(3): 158-161 ambiphilic character the electronic properties and reactivity of radicals are tunable and these species display reactivity towards both electron-rich and electron-poor substrates. As such polar effects play an important role in the reactivity of radicals allowing for practical strategies such as Polarity Reversal Catalysis. Thus radicals can be stabilized by both electron-withdrawing and electron-donating substituents. The electronic structure of super-stable radicals such as O2 nitric oxide and TEMPO reveals the importance of the stabilizing interaction between the SOMO and the adjacent lone pair. Such strong 2-center 3-electron (2c-3e) interactions correspond to the bond order of ½ between the radical center and the heteroatom and have contributed to interesting transformations [26]. In captodative radicals the radical center is flanked by substituents of opposite polarity (i.e. a donor and an acceptor). Such radicals [27] enjoy a special stabilization the extent of which is still under debate [28]. The nature of stabilizing interactions in radical species can extend beyond directly adjacent substituents. In through-bond (TB) interactions spatially separated orbitals interact through their mutual mixing with the σ orbitals of the intervening framework. Such electronic effects are well-known when both of the non-bonding orbitals are singly occupied as in 1 4-diradicals. In these systems TB coupling of radical centers [29] increases the population of the σ* bridge orbital ultimately leading to fragmentation into two double bonds. The same effect is responsible for rendering the Bergman cyclization a symmetry- allowed reaction [30] and providing about 3–5 kcal/mol stabilization to p-benzyne [31]. Recently [32] the same TB interaction was shown to stabilize monoradicals. In this case TB communication between the monoradical and a lone pair three bonds away distinctly stabilizes the radical center as shown in Figures 3 and 4. 3. DETECTION AND CHARACTERIZATION The detection and characterization of radical species are essential for full understanding their reactivity. Nevertheless due to their transient nature and corresponding short life-times characterization of radical species is difficult. First observed in 1994 electron paramagnetic resonance (EPR) spectroscopy has been a primary method for the detection paramagnetic species. If the half-life of the respective radical is too brief for detection spin traps (commonly nitrone derivatives) are used to trap the radical converting it into a stabilized detectible radical adduct. Time-resolved EPR and NMR techniques like chemically induced dynamic electron polarization has allowed real time observation of radicals and radical ions at the nanosecond-to-microsecond time scale elucidating structural details and insights into reaction kinetics. Developed by Comisarow and Marshall in 1974 [33] Fourier transform ion cyclotron resonance mass spectrometry has been the standard Scheme 1: History of radical reactions. Scheme 2: The ambiphilic nature of radical species originates in the net stabilizing SOMO- lowest unoccupied molecular orbital as well as SOMO-highest occupied molecular orbital interactions. Figure 1: The diversity of radical species. Figure 2: Examples of π and σ-radicals.References
No references available for this article.
Citation
Sayantan Mondal*. Structure and Electronic Features of Radicals: Detection and Characterization. Indian J. Adv. Chem. Sci. 2023; 11(3):158-161.