Analytical Method Validation for Quantitative Estimation of Fenoterol-Hydrobromide by Reversed-Phase High-Performance Liquid ChromatographyMethod
B. Ramakrishna*, N. Venkatasubba Naidu, N.S. Hanumantha Rao, B. Vijaya Kumar Naidu*
DOI: 10.22607/IJACS.2023.1103007
Volume 11, Issue 3 | Pages: 209-216
Abstract
A simple, accurate, and precise reverse-phase high-performance liquid chromatographic method was developed for the
estimation of fenoterol hydrobromide in pharmaceutical formulations. A mixture of 70 volumes of buffer solution was prepared
by adding 1 ml of triethylamine in 1000 ml of water adjust to pH = 5.0 with formic acid, and 30 volumes of acetonitrile at a
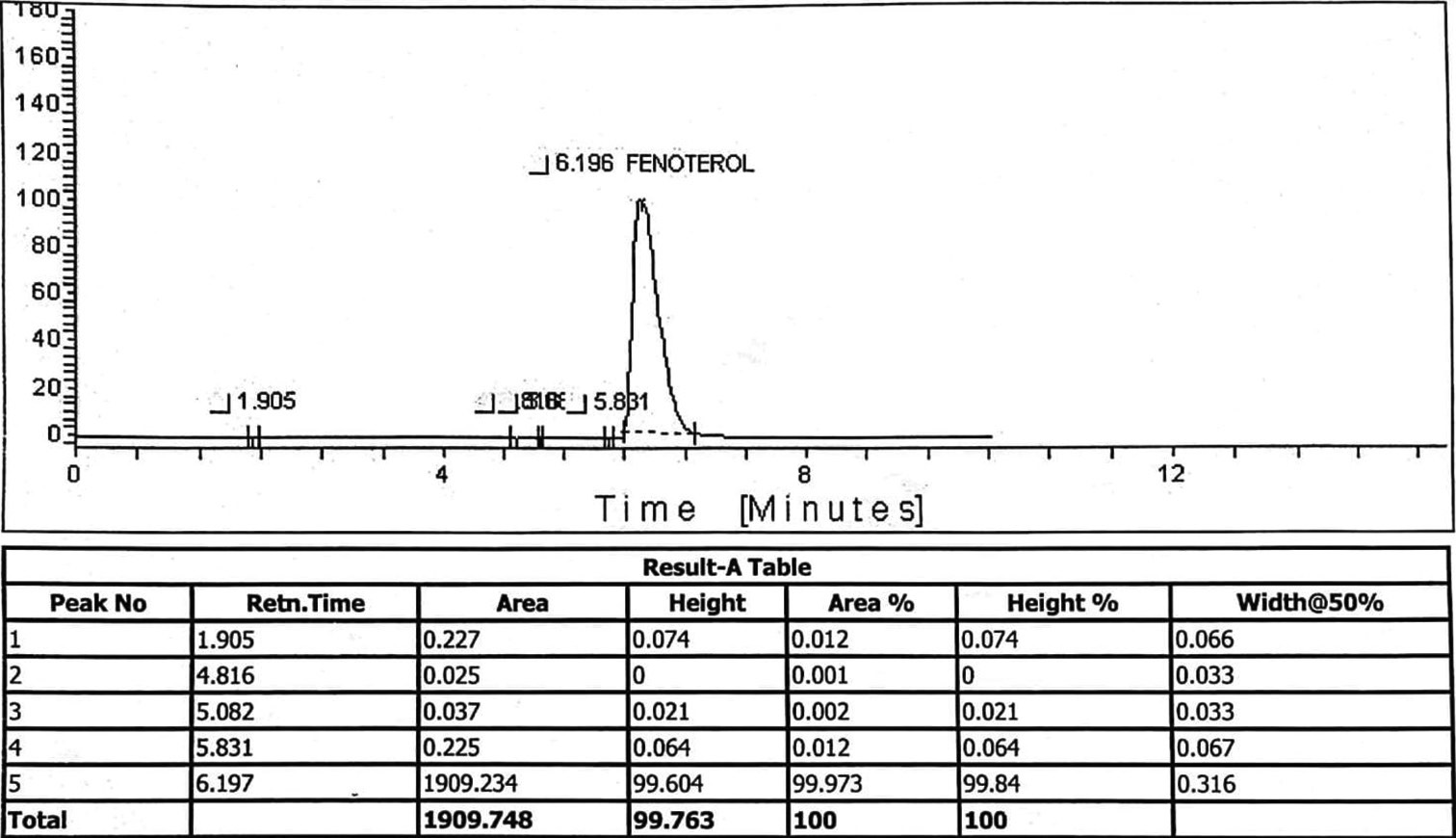
flow rate of 1.0 ml/min with UV detection at 276 nm. The concentration range was from 50% to 150%, the retention time was
6.163 min and the correlation coefficient of the analytical curve was 0.999. The limit of detection and limit quantification were
0.002 mg/mL and 0.006 mg/mL, respectively. Intra- and inter-day relative standard deviations were ≤2.0%. The methodology
accuracy showed the percentage between 98.61% and 101.11%. The described technique was found to be simple, rapid, precise,
accurate, and sensitive; the advantages over the other current methodologies are the low-cost and low-polluting conditions.
Owing to its simplicity and reliable results, this methodology is suitable to be used in the quality control of pharmaceutical drugs
containing fenoterol hydrobromide as an active component.
Keywords
Fenoterol hydrobromide High-performance liquid chromatography Stability indicating studies Pharmaceutical drug validation.References
No references available for this article.
Citation
B. Ramakrishna*, N. Venkatasubba Naidu, N.S. Hanumantha Rao, B. Vijaya Kumar Naidu*. Analytical Method Validation for Quantitative Estimation of Fenoterol-Hydrobromide by Reversed-Phase High-Performance Liquid ChromatographyMethod. Indian J. Adv. Chem. Sci. 2023; 11(3):209-216 .